
Mrs Naiyana Wattanasri, the outgoing Chairman and Founder of the Thailand Medical Technology Council, helped develop a systems quality roadmap for laboratory accreditation for the entire country. In this exclusive interview with Roche Diagram magazine, she speaks about the Laboratory Benchmarking Survey and experiences improving laboratory performance in Thailand.
When Mrs Wattanasri looks back on her experience of over 40 years in laboratory quality management, her proudest achievement is establishing the Thailand Medical Technology Standard in 1998. This accreditation pathway helped align laboratory processes to local needs.
“When we were designing the accreditation process, I questioned the suitability of importing wholesale the western standards and instead established a suitable standard that every laboratory in Thailand could use.”
Mrs Wattanasri explains that while western laboratories tend to separate administrative functions from technical ones, she saw the rationale for merging both. The goal was to create greater synergies between quality process management staff and those responsible for laboratory safety.
This approach was commended by the World Health Organisation (WHO) for helping to reduce gaps in existing laboratory standards and those stipulated by ISO. Thailand’s success is now enabling other countries across Asia, as well as Africa, to adopt a similar approach. Today, the perception of the laboratory’s role has shifted significantly with laboratories firmly established at the core of an increasingly complex healthcare system, according to Mrs Wattanasri.
“Physicians, nurses and patients all see the importance of the laboratory test results and the value we can bring,” she said.
A first for Thailand: Benchmarking to improve laboratory quality, speed and efficiency
The laboratory is crucial to the diagnostic cycle and therefore needs to have a programme in place to monitor the diagnostic process. This can help identify, address and reduce errors while boosting efficiency. Studies show that benchmarking exercises to analyse performance against other laboratories provide a scientific way of evaluating gaps and instituting continuous improvement of common laboratory tasks and functions.
The need for benchmarking is especially acute in Asia Pacific, where there is an urgency around delivering better diagnosis and treatment to vast populations. Since 2011, Roche has been conducting surveys to understand the state of laboratory medicine in the region. The idea was to introduce benchmarking of certain key quality indicators to a wide range of laboratories. The questionnaires were designed to elicit information on three key areas of quality, speed and cost with a focus on clinical chemistry and immunoassays. It has now evolved into one of the largest surveys of its kind in the Asia Pacific region, with the 2015 survey drawing 643 participants across 13 countries.

Three years ago, the Thailand Medical Technology Council partnered with Roche Diagnostics to run the National Laboratory Benchmarking survey. Mrs Wattanasri said it was the first time the Council collaborated with a healthcare company to conduct the survey with a view to bring a new level of insight into process improvements.
“We decided to go with Roche because this is a company that also provides academic expertise to its customers. We reviewed previous surveys and saw that the results were very valuable to laboratories,” she added.
In order to tailor the survey to the local context, Mrs Wattanasri said the Council added questions about the problems faced by laboratories in their daily operations, ranging from procurement systems to internal quality controls, and laboratory safety systems.
The Council introduced the survey during the Laboratory Accreditation Forum 2014, an annual meeting for medical laboratories in Thailand. Over 300 participants from across the country were encouraged to join and more than half of them did so voluntarily. With 155 participating hospitals, the Laboratory Benchmarking Survey provided useful insights into local practices and ways to improve performance.
According to Mrs Wattanasri, the survey results highlighted that the participating laboratories had the expected levels of management but were facing some key challenges.
Results showed that 42% of laboratories had international accreditation with ISO 15189 being the most common type of accreditation. External Quality Checks (EQC) were considered important and 57% of the participating laboratories said they were involved in such programs.
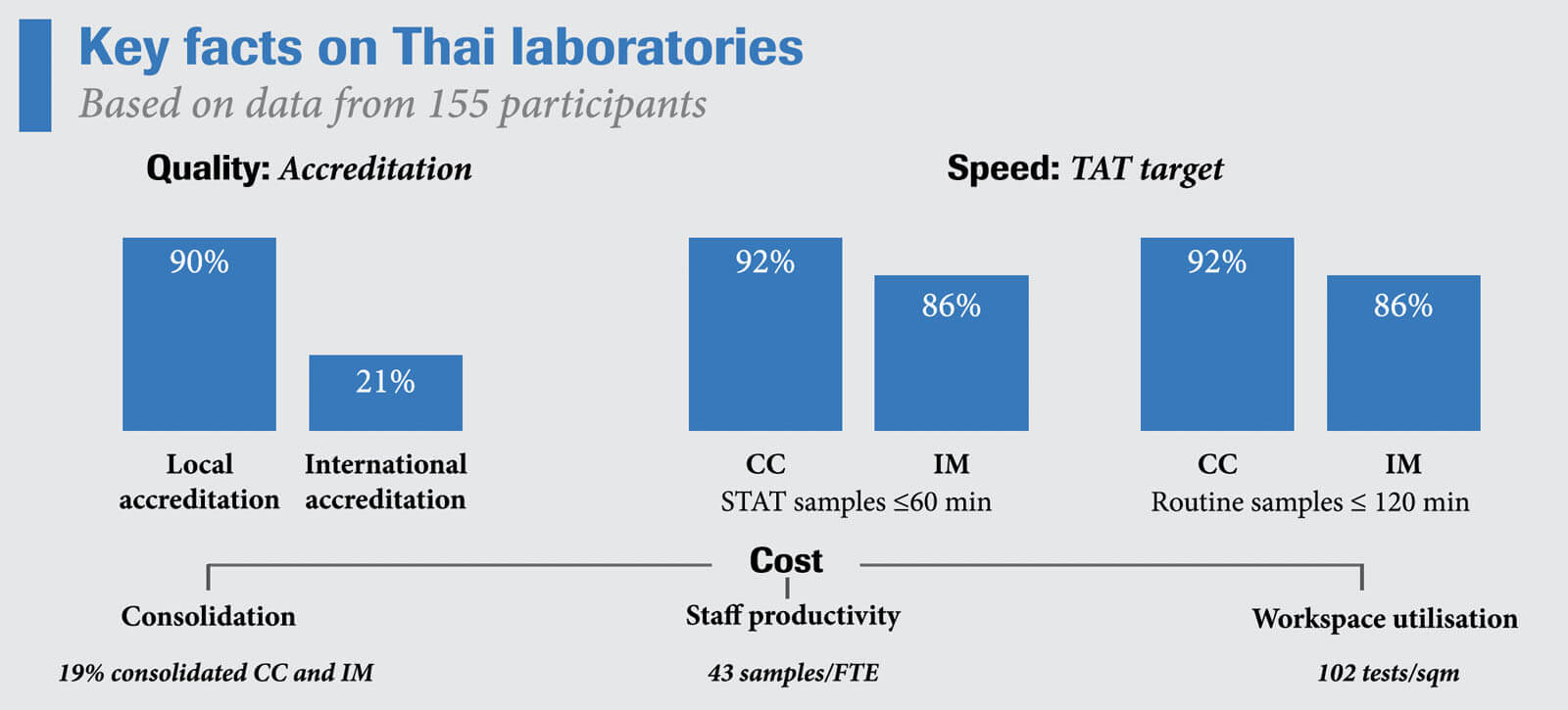
Another aspect examined by the survey was the turn-around time (TAT). Each laboratory sets its own key performance indicators of TAT for each step of the whole testing process, including pre-analytic TAT, analytic TAT and post-analytic TAT. The survey results showed that most errors occurred during the pre-analytic phase. “Around 10% of participants had automated pre-analysis and more were encouraged to do so to reduce errors,” Mrs Wattanasri said. Another challenge the survey highlighted was the lack of human resources, which affected the speed with which results were released to physicians. Though many laboratories rely on their information technology (IT) systems for auto validation, haematology and clinical chemistry test results require manual approval.
Mrs Wattanasri noted that the lack of trained personnel, limited budgets and inconsistent quality standards are challenges common to laboratories across the region. For instance, only 63% of participants said they complete internal quality controls due to budgetary limitations.
“We shared the results with 50 laboratory networks across the country so that they could benchmark themselves according to the results,” she added.
Mrs Wattanasri said the council also shared the survey results with various stakeholders including the Ministry of Public Health and the National Health Security Office so that funding and purchasing directives could be better tailored to support laboratory needs. Recently, the Ministry of Health issued a new code guiding laboratories on how to select and use new medical equipment more efficiently.
Two years after the survey, the yearly customer satisfactory survey conducted by the laboratory networks shows positive sentiment among physicians and patients.

We shared the results with 50 laboratory networks across the country so that they could benchmark themselves according to the results.
Some of post-survey continuous improvement activities conducted by the participants are accreditation, Lean Six Sigma, customer (physicians, patients and other healthcare personnel) satisfactory survey, continuous training for employees, and error rate monitoring.
As an industry veteran, Mrs Wattanasri’s advice to aspiring laboratory technicians is to have a patient-focused mindset.
“When you see patients you can tell they are anxious about their lab results, as it guides the physicians’ decision-making. So your job as a laboratory technician is no longer relegated to the background, it is at the core of the patient’s journey.”














