Dr Thomas C. Wright Jr, Professor Emeritus of Pathology and Cell Biology at Columbia University is actively involved in the study of the pathogenesis of cervical cancer and novel approaches to cervical cancer prevention. He has been on expert panels for the World Health Organization, National Institutes of Health, American Cancer Society and the Centers for Disease Control and Prevention. In this exclusive interview with Dia:gram, Wright discusses the potential of vaccination and new screening technologies to prevent and ultimately eradicate cervical cancer.
When it comes to cervical cancer, there is today an unprecedented opportunity to detect and prevent cancer even before it develops and save lives. Yet, cervical cancer remains one of the most common and deadliest cancers affecting women across the world. In less developed regions, cervical cancer is the second most common cancer in women accounting for 84% of the total cases worldwide in 2012.1 In the same year, more than 85% of the approximately 270,000 cervical cancer deaths occurred in low- and middle-income countries in Asia.1 This is especially tragic as the disease tends to strike women in their prime – when their families are still young and when many of them are trying to establish themselves in their careers. So it also has a huge social and economic impact on the community at large.
Dr Wright who is one of the world’s leading experts on cervical cancer, believes that with the vaccines and screening technologies we have today, this disease does not belong on the top 10 list of cancers affecting women. “Even in countries where it is number 10, it should be number 20,” he says.
Why? Two influential discoveries have changed the course of cervical cancer and substantially reduced its burden. The Pap test discovered in 1923 by Dr George Papanicolaou, made it possible to detect abnormal cells in the cervix. Then, in the 1970s a German scientist made the Nobel-prize winning discovery that the human papillomavirus (HPV) types 16 and 18 play a critical role in the development of many cervical cancers. Today, we know HPV 16 and 18 are responsible for over 70% of cervical cancer cases.2 This discovery paved the way for development of the first vaccine to prevent cervical cancer.

Dr. Wright says that vaccination will be able to prevent and eradicate cervical cancer globally only in combination with screening and HPV genotyping. “Even with vaccination, it’s unlikely we’re going to get everybody vaccinated. So we do need to screen people two or three times during their lives. We therefore need the most sensitive possible screening in that situation,” he says.
There is an unprecedented opportunity to detect cancer even before it develops and save lives.
That cervical cancer screening is now available: In 2011, the United States’ Food and Drug Administration (FDA) approved the first HPV DNA Test that allows HPV 16 and HPV 18 genotyping concurrently.3 The approval was based on the ATHENA trial,4 a multicenter, prospective study supported by Roche, which enrolled over 47,000 women above 21 years of age undergoing routine cervical cancer screening. Results from the ATHENA trial revealed that one in 10 women who tested positive for HPV 16/18 had high-grade cervical disease that was missed by the Pap test and that after 3 years of follow-up one in four women with HPV 16 had developed high-grade cervical disease.5 The trial evaluated the performance of the HPV DNA test in multiple clinical situations including atypical squamous cells of undetermined significance (ASC-US) triage and co-testing. The trial showed that the HPV DNA test increases the detection rate to about 93 to 95% of high-grade precursors.3 Cervical cancer screening guidelines in the United States now recommend HPV co-testing to women aged 30 and above.6
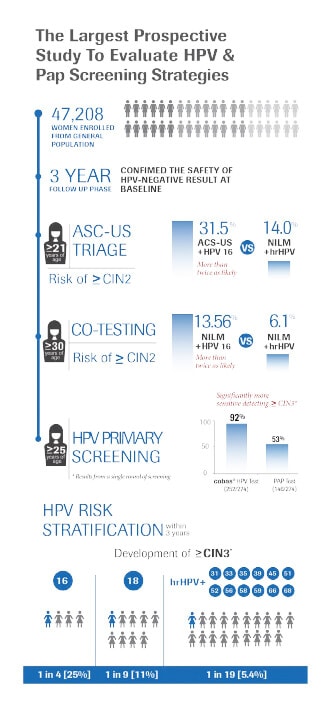
For many countries in the world today, the Pap test is still the primary screening tool in detection of cervical cancer and continues to be valuable in the fight for cervical cancer prevention. But it is not without its limitations. Many studies have confirmed that the sensitivity of the Pap test is rather poor at about 52%.7 It is a subjective test and it can sometimes provide inconclusive results that may lead to unnecessary interventions and stress for the woman.
In contrast, HPV testing and genotyping provides a higher level of objectivity and specificity.8 “HPV primary screening is now going to be the way Australia screens. The Netherlands will also be implementing HPV primary screening. The United Kingdom has recently decided based on the results of a large pilot study to implement HPV primary screening. And there are a number of other countries which are getting ready to do it. In Italy, nine regions have adopted HPV primary screening,” Dr Wright says.
In South East Asia, a pilot co-testing program run out of Singapore General Hospital has shown that cervical cancer can be tested for with greater accuracy by combining Pap smear (cytology) and a validated HPV DNA test.9 The program aimed to improve the detection rate of high grade cervical disease (CIN3+) and reduce the number of screening tests during a woman’s lifetime.
Dr Wright first started talking about HPV in 1995, particularly about the clarity it could bring when Pap results were inconclusive. “No one wanted to do HPV in ’95. We got it accepted through an incremental approach,” he adds. Dr Wright’s decision to become a gynaecologic pathologist was in part fueled by the discovery of the HPV tumour virus. “HPV was becoming important at that point. The first HPV capture version, and we started running clinic trials, and I’ve been working on HPV ever since,” he says.
Since the early 2000s, Dr Wright has been working to bring together societies and organizations to push the model of consensus guidelines for the screening and management of cervical diseases. This is an important part of his legacy as is the work he has put in to developing cervical cancer screening programs.
“I’ve been working for almost 20 years in Sub-Saharan Africa, trying to develop screen and treat approaches for people who don’t have access to what we consider routine cervical cancer screening. The next step is to implement what we’ve learned, in terms of vaccination and HPV testing, in order to be able to prevent cervical cancer where the burden is greatest.”
Dr Wright says one of the most important tasks is to get cervical cancer screening on national healthcare agendas: “Even if globally we were able to get high vaccination coverage of girls, you’ve got a whole population of women who are older than the vaccination age who will need screening. Vaccination is great for preventing cancer 20 or 30 years from now, but you don’t want to tell a whole generation of women that they don’t have prevention.”
*The information contained in this article was extracted from Edition 2017, Vol 1.