Until recently, the world seemed to be making progress against some of the most persistent infectious diseases. In 2019, 59 percent fewer people died from HIV-related causes as compared to 2004, and 37 percent fewer than in 2010.1 However, that progress now appears to be stalling. Only three countries in Asia Pacific — Australia, Cambodia and Thailand — are on track to achieve the United Nations (UN) ‘90-90-90’ target for HIV in 2020.2
Other diseases are adding to the current burden. According to the World Health Organization (WHO), 1.4 million people died from tuberculosis (TB) globally in 2019 (208,000 of whom also had HIV).3 India had the highest number followed by China and Indonesia.3 Meanwhile, Hepatitis B (HBV) is responsible for more than 800,000 deaths globally4 every year. According to WHO’s 2016 estimates, some 3.2 percent of the global population is infected with chronic HBV, and almost a third live in China.5 In all these cases, ensuring more people are diagnosed can stop the spread.

A Domino Effect of COVID-19 on Infectious Disease Management
The current pandemic has put pressure on health services throughout the world. In particular, it has magnified and highlighted existing problems in developing countries, where poor medical infrastructures and insufficient medical staff may lead to inadequate screening.
Despite the need to modernise, many countries are failing to implement the necessary policies to put in place adequate financing, not least for diagnostics. When the system fails to support testing the knock-on effect is clear: more people stay unaware of their HIV status. An opt-out approach to diagnosis is an approach that has been tested in certain countries with successful results, uncovering swathes of people who would otherwise have gone undiagnosed.6 Such an approach could fold in other diseases, thereby boosting detection numbers for TB and hepatitis allowing for efficient clinical decision-making and patient care, especially if diagnosed at earlier stages of their illness.6 With an effective testing and follow-up treatment plans in place, such programmes could reduce the rate of transmission of these diseases in the community.
When the system fails to support testing the knock-on effect is clear: more people stay unaware of their HIV status.
There are good reasons to synthesise the fight against these three diseases into one. Failure to offer a holistic approach to healthcare, including early screening, can lead to the late detection of comorbidities. This kick-starts another chain reaction. A compromised immunity from prior infections leads to more patients with comorbidities, who then need complex clinical management.7 This further exacerbates the pressure on healthcare resources. HIV, HBV and TB patients can also develop antimicrobial resistance (AMR), making them harder and costlier to treat.7 The more you can open access to testing, the less chance there is of people developing multiple infections.
There is another way patient access can be supported — innovation in test kits. For example, plasma separation technology, which protects samples even in heat and humidity, ensures that samples can be transported from rural areas to reliable testing labs many miles away. Similarly, techniques such as Dried Blood Sampling diagnostics enable infants to be HIV tested in a non-invasive manner and eliminate the need for sample refrigeration. Smoothening the process of infant testing is key, as early diagnosis means they can receive care before symptoms develop. Advancements like this are just the tip of the iceberg, but for progress to continue, so must innovation. Continual improvements that make testing easier and cheaper will help boost the number of people given a new lease of life.
Related read: The Emerging Importance of Microbiology Diagnostics
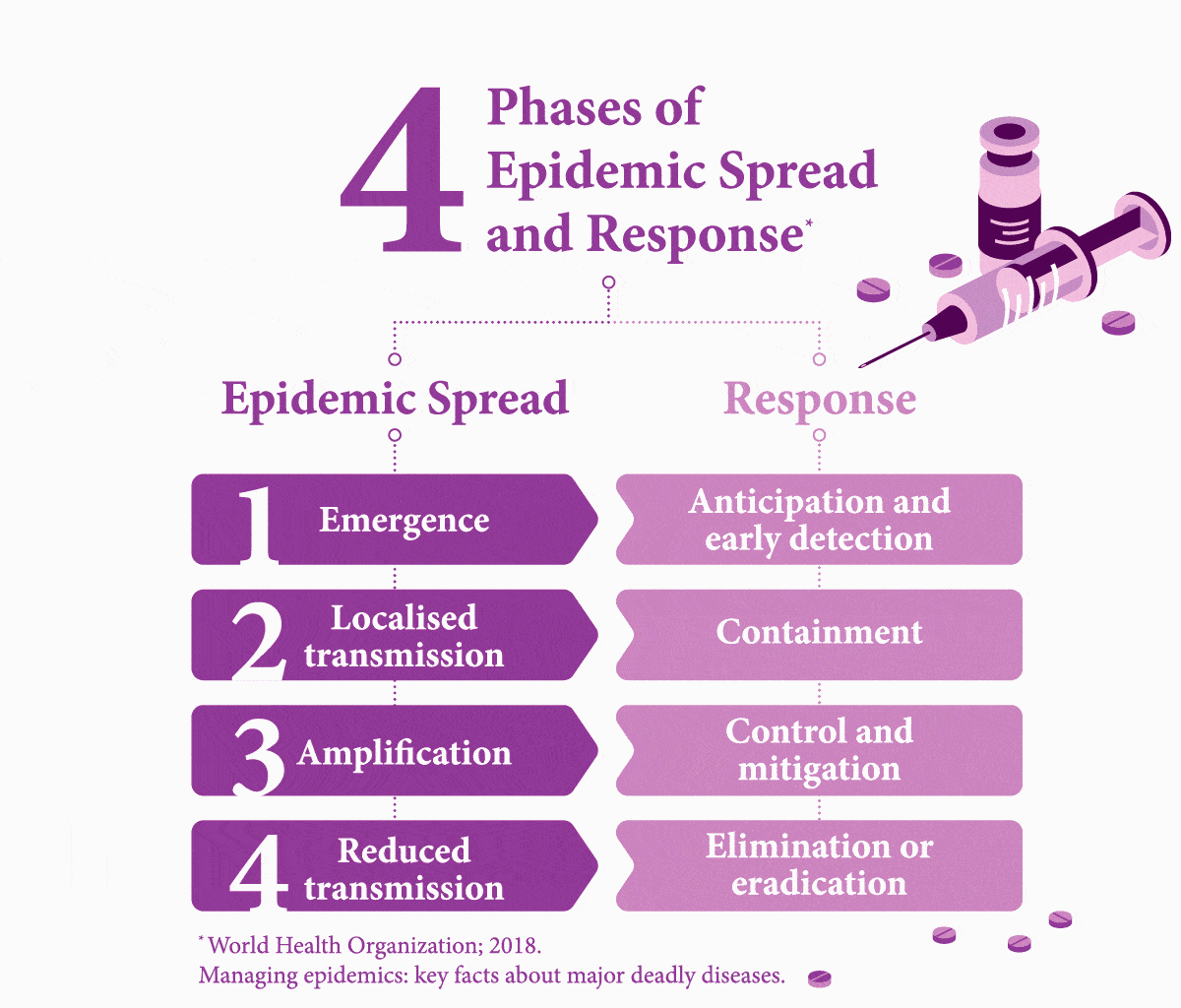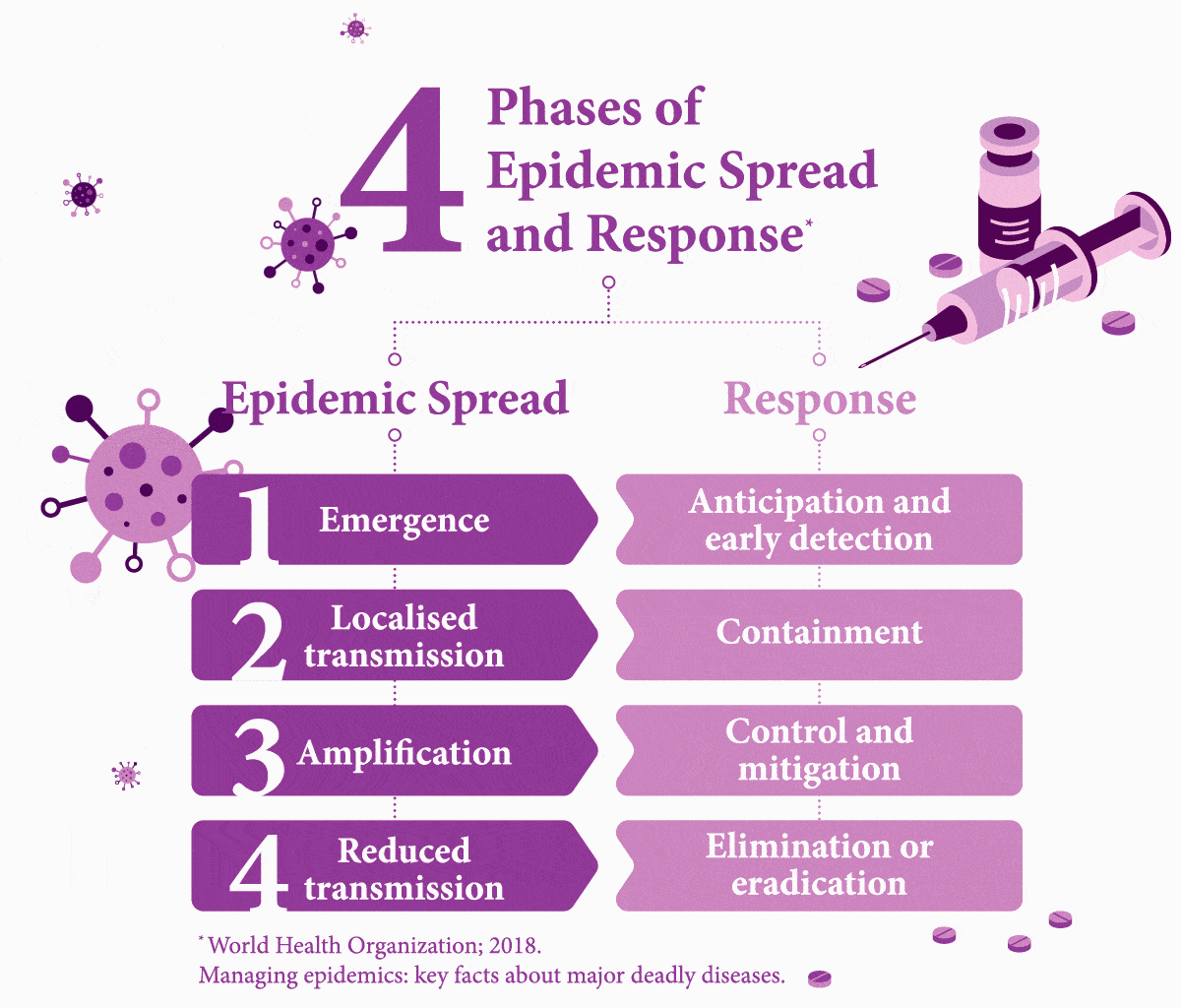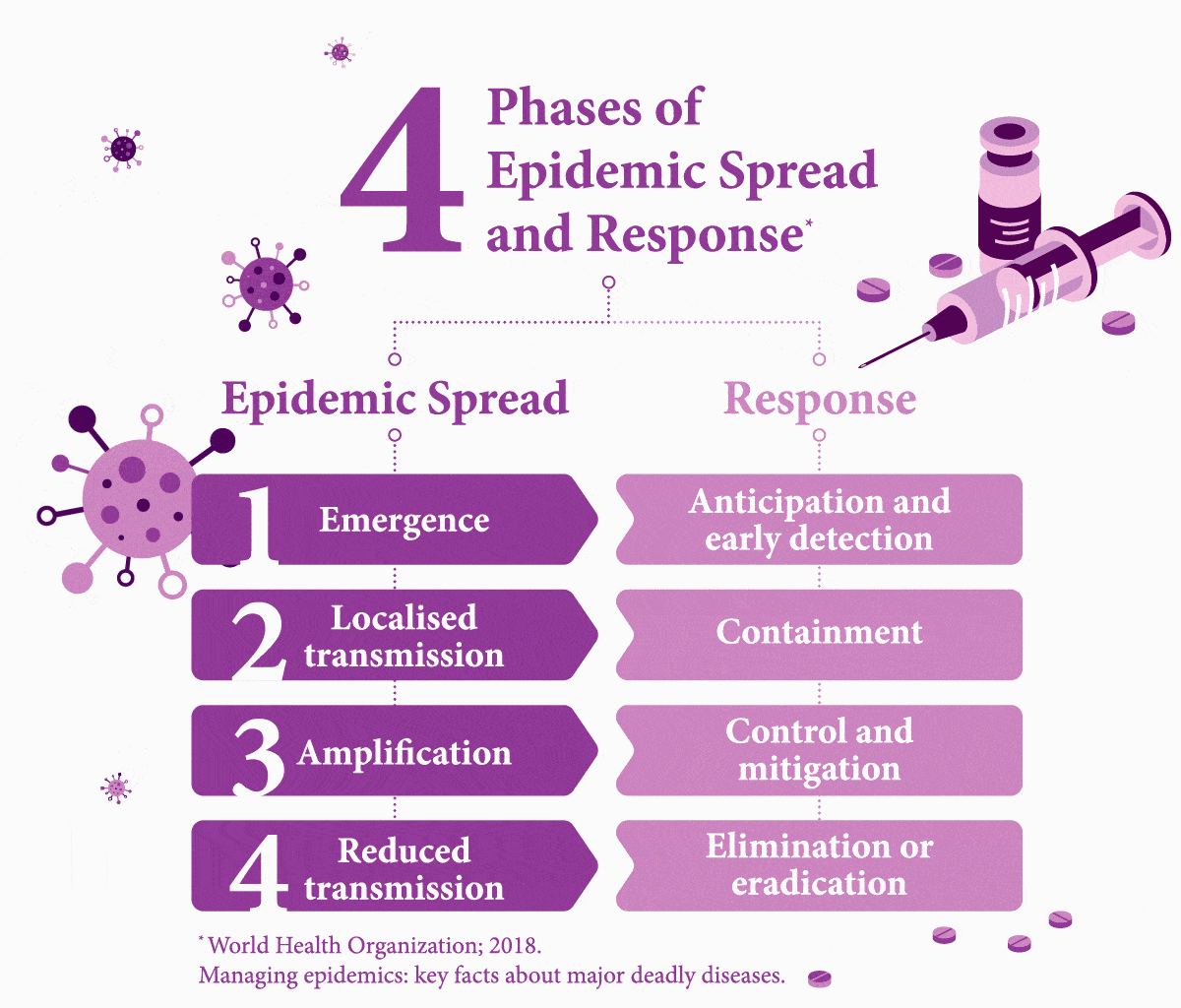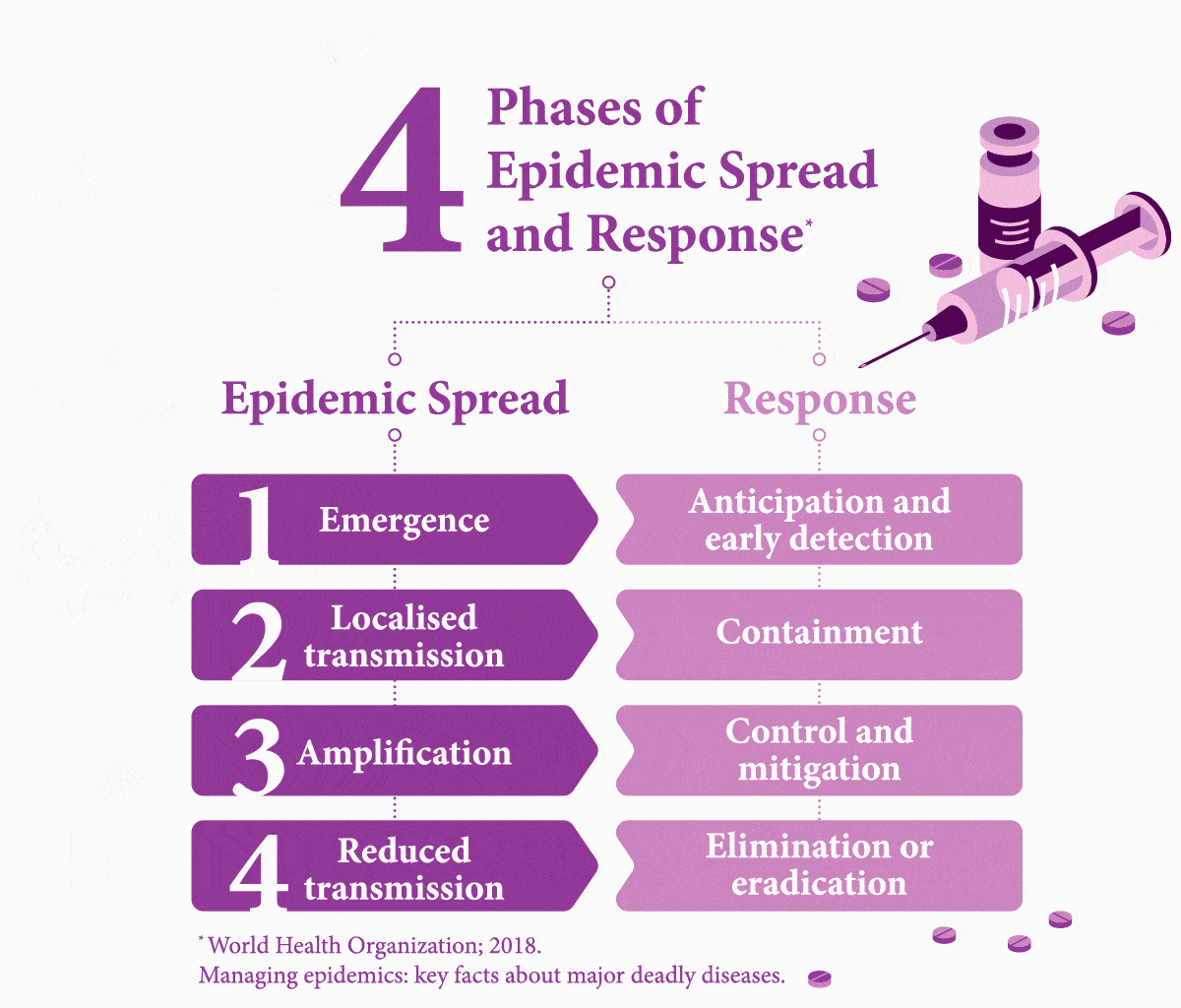
Early Testing is Key. What Stands in the Way?
It is easier to treat a patient for one condition before their weakened immune system makes them vulnerable to further disease — but first they need to be tested. Nine percent of HIV-assisted TB cases in the world are found in India, where it causes 11,000 deaths each year.8 About 10 percent of TB patients in India die due to the lack of access to adequate diagnostic and treatment facilities.9 While developing infrastructure to fill these gaps in healthcare would cost an estimated US$290 million, this is only a fraction of the cost (US$32 billion) of mortality from TB alone.9
Worldwide, it is estimated that 49 percent of people with both diseases are unaware of their co-infection due to not being tested,10 and therefore fail to receive the necessary care.
Another challenge to diagnostics is painfully simple: distance. In emerging countries like India, testing centres are more prevalent in cities. Simply reaching a test centre can be difficult or costly for poorer people in rural areas,11 even if the test itself is subsidised. Simple measures, however, such as providing transport subsidies could boost access to testing, as noted in a study of low-income rural TB patients in China.12 A 2017 report on a survey that focuses on viral hepatitis testing in low- and middle-income countries, including Southeast Asia, noted that it was common for patients to have to foot the bill — another barrier to getting people diagnosed.13 “In terms of future policy,” the report explained, “there is scope for involving non-health workers with task-shifting to promote testing as achieved with HIV.” The authors added that increased testing requires multiple factors that need to combine, such as bolstered community awareness, healthcare worker training, building robust national strategies, and better access. Because to make headway in managing these infectious diseases effectively, governments must build an interlinked chain reaction of success.

Big Targets, Better Collaboration
It is clear that a coherent strategy for diagnosis and treatment needs to be in place to improve healthcare access. The WHO states that “we need to improve data systems, diagnostics, laboratories and drug supply, and provide better linkages with other services […] using integrated service delivery models such as ‘one-stop shops’.”10
Furthermore, the WHO aims for a 90 percent reduction in new chronic hepatitis infections.14 The target for TB is an end to the disease by 2035.15 To achieve this, however, testing needs to come to the forefront, by identifying new cases and starting the patient journey towards care.
Goals like this are ambitious but crucial. They provide the urgent spur the world needs, if we are to start controlling persistent infectious diseases.
Read more healthcare and diagnostics news at Roche publications.
References:
1World Health Organization. (2020). Number of deaths due to HIV/AIDS
Estimates by WHO region. Global Health Observatory data repository. Retrieved from https://apps.who.int/gho/data/node.main.623?lang=en
2Joint United Nations Programme on HIV and AIDS. (2020). Global Aids Update 2020 — Seizing the Moment: Tackling entrenched inequalities to end epidemics. Retrieved from https://www.unaids.org/sites/default/files/media_asset/2020_global-aids-report_en.pdf
3World Health Organization. (2020). Tuberculosis. Retrieved from https://www.who.int/news-room/fact-sheets/detail/tuberculosis
4Liu J, Liang W, Jing W, and Liu M. (2018). Countdown to 2030: eliminating hepatitis B disease, China. Bulletin of the World Health Organization 2019;97:230-238. http://dx.doi.org/10.2471/BLT.18.219469
5World Health Organization. (2016). Up to 10 million people in China could die from chronic hepatitis by 2030 – Urgent action needed to bring an end to the ‘silent epidemic’. Retrieved from https://www.who.int/china/news/detail/26-07-2016-up-to-10-million-people-in-china-could-die-from-chronic-hepatitis-by-2030-urgent-action-needed-to-bring-an-end-to-the-silent-epidemic-
6O’Connell S, Lillis D, Cotter A, et al. (2016). Opt-Out Panel Testing for HIV, Hepatitis B and Hepatitis C in an Urban Emergency Department: A Pilot Study. PloS one, 11(3), e0150546. https://doi.org/10.1371/journal.pone.0150546
7Unitaid. (2018). Multi-disease Diagnostic Landscape for Integrated Management of HIV, HCV, TB and Other Coinfections — January 2018. Retrieved from https://unitaid.org/assets/multi-disease-diagnostics-landscape-for-integrated-management-of-HIV-HCV-TB-and-other-coinfections-january-2018.pdf
8Central TB Division, Ministry of Health and Family Welfare, Government of India. (2019). India TB Report 2019 — Revised National TB Control Programme Annual Report. Retrieved from https://tbcindia.gov.in/WriteReadData/India%20TB%20Report%202019.pdf
9Reid M, Arinaminpathy M, Bloom A, et al. (2019). Building a tuberculosis-free world: The Lancet Commission on tuberculosis. The Lancet Commissions, 393;10178:1331-1384. https://doi.org/10.1016/S0140-6736(19)30024-8
10World Health Organization. (2018). TB causes 1 in 3 HIV deaths. Retrieved from https://www.who.int/hiv/mediacentre/news/hiv-tb-patient-centred-care/en/
11Subbaraman R, Thomas BE, Sellappan S, et al. (2017). Tuberculosis patients in an Indian mega-city: Where do they live and where are they diagnosed?. PLoS One. https://doi.org/10.1371/journal.pone.0183240
12Zhao Q, Wang L, Tao T, and Xu B. (2013). Impacts of the “transport subsidy initiative on poor TB patients” in Rural China: a patient-cohort based longitudinal study in rural China. PloS one, 8(11), e82503. https://doi.org/10.1371/journal.pone.0082503
13Ishizaki A, Bouscaillou J, Luhmann N, et al. (2017). Survey of programmatic experiences and challenges in delivery of hepatitis B and C testing in low- and middle-income countries. BMC infectious diseases, 17(Suppl 1), 696. https://doi.org/10.1186/s12879-017-2767-0
14World Health Organization. (2020). Hepatitis. Retrieved from https://www.who.int/health-topics/hepatitis
15World Health Organization. (2015). WHO End TB Strategy — Global strategy and targets for tuberculosis prevention, care and control after 2015. Retrieved from https://www.who.int/tb/post2015_strategy/en/
16World Health Organization. (2016). Combating Hepatitis B and C to Reach Elimination by 2030. Retrieved from https://apps.who.int/iris/bitstream/handle/10665/206453/WHO_HIV_2016.04_eng.pdf
17Roche. (2020). Global Access Program. Retrieved from https://www.roche.com/sustainability/access-to-healthcare/global-access-program.htm













